
^ "Inorganic and Organic Lead Compounds" (PDF).International Labour Organization, International Occupational Safety and Health Information Centre. ^ "Lead nitrate, Chemical Safety Card 1000"."(S)-N-(9-Phenylfluoren-9-yl)alanine and (S)-Dimethyl-N-(9-phenylfluoren-9-yl)aspartate". ^ "Auxiliary agents in gold cyanidation".Revisa de la Facultad de Ingeniera, Universidad Central de Venezuela. "2D 4.8 2 Network with threefold parallel interpenetration from nanometre-sized tripodal ligand and lead(II) nitrate". ^ Shuang-Yi Wan Jian Fan Taka-aki Okamura Hui-Fang Zhu Xing-Mei Ouyang Wei-Yin Sun & Norikazu Ueyama (2002).Heating lead nitrate is convenient means of making nitrogen dioxide 2 Pb ( NO 3 ) 2 → Δ 2 PbO + 4 NO 2 + O 2 ". Lead nitrate has been used as a heat stabiliser in nylon and polyesters, as a coating for photothermographic paper, and in rodenticides. This is potentially due to a lead lone pair of electrons, also found in lead complexes with an imidazole ligand. One interesting aspect of this type of complexes is the presence of a physical gap in the coordination sphere i.e., the ligands are not placed symmetrically around the metal ion. The crystal structure shows that the nitrate group forms a bridge between two lead atoms. The complex formed by lead nitrate with a bithiazole bidentate N-donor ligand is binuclear. The total coordination number is 10, with the lead ion in a bicapped square antiprism molecular geometry. The two bidentate nitrate ligands are in trans configuration. In the crystal structure for this compound, the EO 5 chain is wrapped around the lead ion in an equatorial plane similar to that of a crown ether. For example, combining lead nitrate and pentaethylene glycol (EO 5) in a solution of acetonitrile and methanol followed by slow evaporation produced the compound. Lead(II) is a hard acceptor it forms stronger complexes with nitrogen and oxygen electron-donating ligands. Solutions of lead nitrate can be used to form co-ordination complexes. There is no evidence for the formation of the hydroxide, Pb(OH) 2, in aqueous solution below pH 12. The cation 4+ is unusual in having an oxide ion inside a cluster of 3 face-sharing PbO 4 tetrahedra. Pb 2(OH) 2(NO 3) 2 is the predominant species formed at low pH. It is soluble in water and dilute nitric acid.īasic nitrates are formed in when alkali is added to a solution. Lead nitrate decomposes on heating, a property that has been used in pyrotechnics. Solubility of lead nitrate in nitric acid at 26 ☌. Solutions and crystals of lead(II) nitrate are formed in the processing of lead– bismuth wastes from lead refineries. It may also be obtained evaporation of the solution obtained by reacting metallic lead with dilute nitric acid. Lead nitrate is produced by reaction of lead(II) oxide with concentrated nitric acid: PbO + 2 HNO 3(concentrated) → Pb(NO 3) 2↓ + H 2O It has been used as an oxidizer in black powder and together with lead azide in special explosives. These pigments were used for dyeing and printing calico and other textiles. It has been produced as a raw material for making pigments such as chrome yellow (lead(II) chromate, PbCrO 4) and chrome orange (basic lead(II) chromate, Pb 2CrO 5) and Naples yellow. It is produced commercially by reaction of metallic lead with concentrated nitric acid in which it is sparingly soluble. Lead nitrate was first identified in 1597 by the alchemist Andreas Libavius, who called the substance plumbum dulce, meaning "sweet lead", because of its taste. Due to its hazardous nature, the limited applications of lead(II) nitrate are under constant scrutiny. Lead(II) nitrate is toxic and must be handled with care to prevent inhalation, ingestion and skin contact. 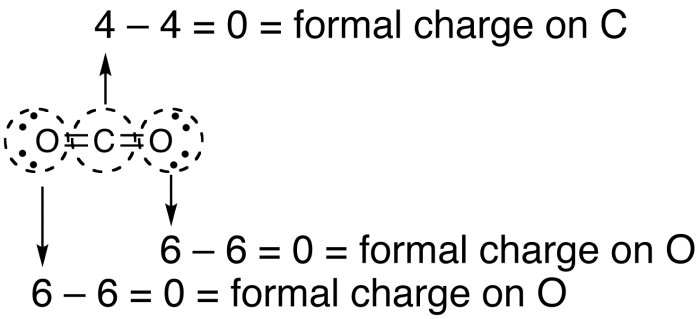
Since around the year 2000, lead(II) nitrate has begun to be used in gold cyanidation. Other industrial uses included heat stabilization in nylon and polyesters, and in coatings of photothermographic paper. Historically, the main use was as a raw material in the production of pigments for lead paints, but such paints have been superseded by less toxic paints based on titanium dioxide. 
In the nineteenth century lead(II) nitrate began to be produced commercially in Europe and the United States. Known since the Middle Ages by the name plumbum dulce, the production of lead(II) nitrate from either metallic lead or lead oxide in nitric acid was small-scale, for direct use in making other lead compounds. It commonly occurs as a colourless crystal or white powder and, unlike most other lead(II) salts, is soluble in water. Lead(II) nitrate is an inorganic compound with the chemical formula Pb( NO 3) 2.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
